What is thermodynamics? Definition, Equations, Laws
Thermodynamics is the study of the relations between heat, work, temperature, and energy. The laws of thermodynamics describe how the energy in a system changes and whether the system can perform useful work on its surroundings.
Basic Theoretical Information
Heat capacity of matter
If as a result of heat exchange a certain amount of heat is transferred to the body, then the internal energy of the body and its temperature change. The amount of heat Q required to heat 1 kg of a substance per 1 K is called the specific heat capacity of the substance c . Then the amount of heat (energy) required to change the temperature of a certain body of mass m can be calculated by the formula:
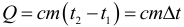
At the same time, in this formula it is absolutely not important in what units the temperature is substituted, since for us, not its absolute value, but change is important. Unit of measurement of the specific heat of the substance: J / (kg ∙ K).
- If t 2 > t 1 , then Q > 0 – the body heats up (receives heat).
- If t 2 < t 1 , then Q <0 – the body is cooled (gives off heat).
The product of body mass and the specific heat of the substance from which it is made is called the heat capacity of the body (ie, simply the heat capacity without the word “specific”):
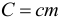
If the problem statement says about the heat capacity of the body, then the amount of heat given or received by this body can be calculated by the formula:

So remember:
- The specific heat is indicated by a small letter c, and is a characteristic of the substance.
- (Simply) The heat capacity is denoted by a large letter C, and is a characteristic of this body.
Recall that the amount of heat Q given away by some source (heater) is calculated by the formula: Q = Pt , where: P is the source power, t is the time during which the source gave off heat. When solving problems, do not confuse time source operation and temperature.
Phase transformations
Phase of substance is called a homogeneous system, for example, a solid body, whose physical properties are the same at all points. Between the various phases of a substance under normal conditions there is a clearly defined boundary (surface) of the section. When the external conditions (temperature, pressure, electric and magnetic fields) change, the substance can move from one phase to another. Such processes are called phase transformations (transitions).
The process of phase transition from liquid to gaseous ( vaporization ) or from solid to liquid ( melting ) can occur only when a certain amount of heat is communicated to the substance. Reverse phase transitions ( condensation and crystallization , or solidification ) are accompanied by the release of the same amount of heat.
The amount of heat entering the system or released from it changes its internal energy. This means that the internal energy of steam at 100 ° C is greater than that of a liquid at the same temperature. These phase transitions occur at constant temperatures, which are called the boiling point and the melting point, respectively. The amount of heat needed to convert a liquid into steam or released by steam during condensation is called the heat of vaporization:

where: r is the specific heat of vaporization . Unit of measurement [ r ] = 1 J / kg. The physical meaning of the specific heat of vaporization: it is equal to the amount of heat needed to convert 1 kg of liquid into a vapor at the boiling point. Turning a liquid into steam does not require bringing the liquid to a boil. Water can turn into steam at room temperature. This process is called evaporation .
The amount of heat needed to melt the body or released during crystallization (solidification) is called the heat of fusion:
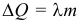
where: λ is the specific heat of fusion . Unit of measure [ λ ] = 1 J / kg. The physical meaning of the specific heat of fusion: the heat required to melt 1 kg of a substance that is at the melting point. The specific heat of vaporization and melting are also called latent heat, since the system temperature does not change during phase transitions, despite the fact that heat is supplied to it.
Note that during phase transitions the temperature of the system does not change. And also the fact that the phase transitions themselves begin only after reaching the required temperature.
The most common source of energy for human needs is fuel – a substance that, when burned, releases a certain amount of heat. The amount of heat released during the combustion of fuel mass m is called the heat of combustion of fuel :
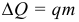
where: q is the specific heat of combustion (calorific value, caloric content) of the fuel. Unit of measure [ q ] = 1 J / kg. The physical meaning of the specific heat of combustion of fuel: a value that indicates how much heat is released during the complete combustion of 1 kg of fuel.
Heat balance equation
In accordance with the law of energy conservation for a closed system of bodies, in which no energy transformations occur, except for heat exchange, the amount of heat given off by more heated bodies is equal to the amount of heat received by colder ones. Heat transfer ceases in the state of thermodynamic equilibrium, i.e. when the temperature of all bodies of the system becomes the same. Let us formulate the heat balance equation: in a closed system of bodies, the algebraic sum of the quantities of heat given and received by all the bodies participating in the heat exchange is zero :
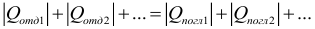
When using this form of writing the heat balance equation, in order not to make a mistake, remember: when you consider heat when heating or cooling a body, you need to subtract less from a higher temperature so that heat is always positive. If all heat is recorded with a sign, where “+” corresponds to the energy received by the body, and “-” to release, then the heat balance equation can be written as:

When using this form of recording, it is always necessary to subtract the initial one from the final temperature. With this approach, the sign of their difference itself “shows” gives the body warmth or receives.
Remember that the body absorbs heat if it occurs:
- The heating,
- Melting,
- Steam formation.
The body gives off heat if it occurs:
- Cooling,
- Crystallization,
- Condensation,
- Combustion of fuel.
It is in this topic, it makes sense not to solve problems in a general form, but to immediately substitute numbers.
Mutual transformations of mechanical and internal energy
With inelastic collisions, the mechanical energy is partially or completely transferred to the internal energy of the bodies, that is, the bodies can heat up and melt. In the general case, the change in mechanical energy is equal to the amount of heat released.
Work perfect gas
Thermodynamics is the science of thermal phenomena. In contrast to the molecular – kinetic theory, which draws conclusions based on the ideas about the molecular structure of matter, thermodynamics is based on the most general laws of thermal processes and properties of macroscopic systems. The conclusions of thermodynamics are based on a set of experimental facts and do not depend on our knowledge of the internal structure of matter, although in a number of cases thermodynamics use molecular – kinetic models to illustrate their conclusions.
Thermodynamics considers isolated systems of bodies that are in a state of thermodynamic equilibrium . This means that all observed macroscopic processes have ceased in such systems. An important property of a thermodynamically equilibrium system is the equalization of the temperature of all its parts.
If the thermodynamic system was exposed to external effects, then eventually it will move to another equilibrium state. Such a transition is called a thermodynamic process . If the process is rather slow (infinitely slow in the limit), then the system at each time instant is close to the equilibrium state. Processes consisting of a sequence of equilibrium states are called quasistatic (or quasistationary , another name for such processes is equilibrium ).
In the isobaric process, the work of an ideal gas can be calculated by the formulas:
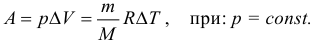
We emphasize once again: gas expansion work can be considered using these formulas only if the pressure is constant. According to this formula, when expanding, the gas performs positive work, and during compression it does negative (that is, the gas resists compression and it is necessary to do work on it in order for it to take place).
If the pressure cannot be considered constant, then the gas operation is found as the area of the figure under the graph in coordinates ( p , V ). Obviously, in the isochoric process, the gas operation is zero.

Due to the fact that the gas operation is numerically equal to the area under the schedule, it becomes clear that the amount of work depends on what kind of process occurred, because each process has its own schedule, and under it its own area. Thus, the work depends not only and not so much on the initial and final states of the gas, but on the process by which the final state was reached.
Internal energy
One of the most important concepts of thermodynamics is the internal energy of the body. All macroscopic bodies have the energy contained within the bodies themselves. From the point of view of the molecular – kinetic theory, the internal energy of a substance is made up of the kinetic energy of all atoms and molecules and the potential energy of their interaction with each other. In particular, the internal energy of an ideal gas is equal to the sum of the kinetic energies of all gas particles that are in continuous and random thermal motion. The internal energy of an ideal gas depends only on its temperature and does not depend on volume. The internal energy of a monatomic ideal gas is calculated by the formulas:

Thus, the internal energy U of the body is uniquely determined by macroscopic parameters characterizing the state of the body. It does not depend on the way in which this state was implemented. It is customary to say that internal energy is a function of state. This means that the change in internal energy does not depend on how the system was transferred from one state to another (it depends only on the characteristics of the initial and final states) and always, in any process for a monatomic ideal gas, is determined by the expression:

Please note: this formula is valid only for monatomic gas, but it is applicable to all processes (and not just to isobar gas, as a formula for work). As can be seen from the formula, if the temperature has not changed, then the internal energy remains constant.
The first law of thermodynamics
If the system exchanges heat with the surrounding bodies and performs work (positive or negative), then the state of the system changes, that is, its macroscopic parameters change (temperature, pressure, volume). Since the internal energy U is uniquely determined by macroscopic parameters characterizing the state of the system, it follows that the heat exchange and work processes are accompanied by a change in Δ U of the internal energy of the system.
The first law (beginning) of thermodynamics is a generalization of the law of conservation and transformation of energy for a thermodynamic system. It is formulated as follows: The change in Δ U of the internal energy of an uninsulated thermodynamic system is equal to the difference between the amount of heat Q transferred to the system and work A performed by the system on external bodies. However, the ratio expressing the first law of thermodynamics is often written in a slightly different form:
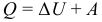
The amount of heat received by the system goes on changing its internal energy and doing work on external bodies (such a formulation is more convenient and understandable, in this form it is quite obvious that this is just a law of energy conservation).
The first law of thermodynamics is a generalization of experimental facts. According to this law, energy cannot be created or destroyed; it is transferred from one system to another and turns from one form to another. An important consequence of the first law of thermodynamics is the statement that it is impossible to create a machine capable of performing useful work without energy consumption from the outside and without any changes inside the machine itself. Such a hypothetical machine was called the perpetuum mobile (perpetuum mobile) of the first kind. Numerous attempts to create such a machine invariably ended in failure. Any machine can perform positive work A on external bodies only by obtaining a certain amount of heat Q from surrounding bodies or reducing Δ U its internal energy.
Adiabatic (adiabatic) is a process in which the system does not exchange heat with the environment. When adiabatic process Q = 0. Therefore: Δ U + A = 0, that is: A = – Δ U . Gas does work by reducing its own internal energy.
The first law of thermodynamics and isoprocesses
For different isoprocesses, formulas can be written out by which the obtained heat Q , the change in internal energy Δ U,and the work of gas A can be calculated . Isochoric process ( V = const):

Isobaric process ( p = const):

Isothermal process ( T = const):
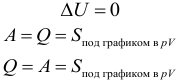
Adiabatic process ( Q = 0):

If the problem does not explicitly state that the gas is monatomic (or one of the inert gases is not named, for example, helium), then the formulas from this section cannot be used.
Cycles Heat Machines
A heat engine is a device capable of converting a quantity of heat into mechanical work. Mechanical work in heat engines is produced in the process of expansion of a substance, which is called the working fluid. Gaseous substances (gasoline vapors, air, water vapor) are commonly used as the working medium. The working body receives (or gives) thermal energy in the process of heat exchange with bodies that have a large supply of internal energy. These bodies are called heat reservoirs.
Really existing heat engines (steam engines, internal combustion engines, etc.) operate cyclically. The process of heat transfer and the conversion of the received amount of heat into work is periodically repeated. For this, the working fluid must perform a circular process or a thermodynamic cycle, during which the initial state is periodically restored.
The common property of all circular processes is that they cannot be carried out, bringing the working fluid into thermal contact with only one heat reservoir. They need at least two. A heat tank with a higher temperature is called a heater , and a lower one is called a refrigerator . Making a circular process, the working fluid receives from the heater a certain amount of heat Q 1 > 0 and gives the refrigerator the amount of heat Q 2 <0.
The efficiency of a heat engine can be calculated by the formula:
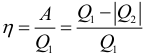
where: Q 1 is the amount of heat received by the working fluid in one cycle from the heater, Q 2 is the amount of heat transferred by the working fluid in one cycle to the refrigerator. Work perfect with a heat machine in one cycle:

Efficiency indicates how much of the heat energy received by the working fluid from the “hot” heat reservoir has turned into useful work. The rest of the (1 – η ) was “useless” transferred to the refrigerator. The efficiency of a heat engine is always less than one ( η <1).
The highest efficiency at given temperatures of the heater T 1 and the cooler T 2 is achieved if the heat engine operates according to Carnot cycle . The Carnot cycle consists of two isotherms and two adiabats. The efficiency of the Carnot cycle is equal to:
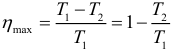
The second beginning (second law) of thermodynamics
The first law of thermodynamics does not establish the direction of the flow of thermal processes. However, as experience shows, many thermal processes can proceed only in one direction. Such processes are called irreversible. For example, with the thermal contact of two bodies with different temperatures, the heat flux is always directed from a warmer body to a colder one. The spontaneous process of heat transfer from a body with a low temperature to a body with a higher temperature is never observed. Therefore, the heat exchange process at a finite temperature difference is irreversible.
Reversible processes are processes of transition of a system from one equilibrium state to another, which can be carried out in the opposite direction through the same sequence of intermediate equilibrium states. In this case, the system itself and the surrounding bodies return to their original state.
The processes of transformation of mechanical work into the internal energy of a body due to the presence of friction, diffusion processes in gases and liquids, gas mixing processes with an initial differential pressure, etc. are irreversible . All real processes are irreversible, but they can come as close as possible to reversible processes. Reversible processes are idealization of real processes.
The first law of thermodynamics cannot distinguish reversible processes from irreversible ones. It simply requires a certain energy balance from the thermodynamic process and says nothing about whether such a process is possible or not. The direction of the spontaneously proceeding processes is established by the second law of thermodynamics. It can be formulated as a ban on certain types of thermodynamic processes.
The English physicist U. Kelvin gave the following wording of the second law in 1851: In a cyclically operating heat engine, a process is impossible, the only result of which would be the transformation into mechanical work of the entire amount of heat obtained from a single heat reservoir.
The hypothetical heat engine in which such a process could take place is called the “perpetual motion machine of the second kind.” As it should have already become clear, the second law of thermodynamics forbids the existence of such an engine.
The German physicist R. Clausius gave a different formulation of the second law of thermodynamics: A process is impossible, the only result of which would be the transfer of energy by heat exchange from a body with a low temperature to a body with a higher temperature. It should be noted that both formulations of the second law of thermodynamics are equivalent.
Difficult problems in thermodynamics
When solving various non-standard problems in thermodynamics, the following remarks should be taken into account:
- To find the work of an ideal gas, it is necessary to plot the process in the coordinates p ( V ) and find the area of the figure under the graph. If a process graph is given in the coordinates p ( T ) or V ( T ), then it is first rearranged to the coordinates p ( V ). If the condition specifies a mathematical relationship between gas parameters, then first find the relationship between pressure and volume, and then build a graph p ( V ).
- To find the work of a mixture of gases using the law of Dalton.
- When combining insulated vessels, the internal energy of the entire system should not change, i.e. how much joule the internal energy of a gas increases in one vessel, how much it decreases in another.
- Generally speaking, the pressure and temperature of a gas can be measured only in the state of thermodynamic equilibrium, when the pressure and temperature are the same at all points of the vessel. But there are situations when the pressure is the same at all points, but the temperature is not. This may be due to different concentrations of molecules in different parts of the vessel (analyze the formula: p = nkT ).
- Sometimes it is necessary to use knowledge from mechanics in problems of thermodynamics.
Calculation of the efficiency of cycles on the schedule
The objectives of this topic are considered to be among the most difficult tasks in thermodynamics. So, to solve, you have to, first, translate the process graph into p ( V ) coordinates. Secondly, it is necessary to calculate the gas per cycle. The useful work is equal to the area of the figure inside the graph of the cyclic process in the coordinates p (V). Thirdly, it is necessary to figure out where the gas is received, and where it gives off heat. To do this, remember the first law of thermodynamics. The internal energy of an ideal gas depends only on its temperature, and the work on the volume. Therefore, gas receives heat if:
- Both its temperature and volume increase;
- The volume increases, and the temperature is constant;
- The temperature increases, and the volume is constant.
Gas gives off heat if:
- Both its temperature and volume decrease;
- The volume decreases, and the temperature is constant;
- The temperature decreases, and the volume is constant.
If one of the parameters increases, and the other decreases, in order to understand, it gives heat to the gas or receives it, it is necessary to calculate the heat and look at its sign “head” on the first start of thermodynamics. Positive heat – gas gets it. Negative – gives.
The first type of tasks. In p ( V ) – coordinates, the cycle graph is a figure with easily calculated area, and the gas receives heat in isochoric and isobaric processes. Apply the formula:

Please note that the denominator is only the heat generated by the gas in one cycle, that is, the heat only in those processes in which the gas received it.
The second type of tasks. In p ( V ) – coordinates, the cycle graph is a figure with easily calculated area, and the gas gives off heat in isochoric and isobaric processes. Apply the formula:

Please note that in the denominator is only the heat given off by the gas in one cycle, that is, the heat is only in those processes in which the gas gave it away.
The third type of tasks. Gas receives heat not in isochoric or isobar processes that are convenient for calculating, there are isotherms or adiabats in the cycle, or “no” processes at all. Apply the formula:

Vapor properties. Humidity
Any substance under certain conditions can be in various aggregative states – solid , liquid and gaseous . The transition from one state to another is called a phase transition . Evaporation and condensation are examples of phase transitions.
Evaporation refers to a phase transition from a liquid to a gaseous state. From the point of view of the molecular – kinetic theory, evaporation is the process by which the fastest molecules fly out from the surface of the liquid, the kinetic energy of which exceeds their binding energy with the rest of the liquid molecules. This leads to a decrease in the average kinetic energy of the remaining molecules, that is, to the cooling of the liquid (if there is no supply of energy from the surrounding bodies).
Condensation is the opposite of evaporation. Upon condensation, the vapor molecules return to the liquid.
In a closed vessel, the liquid and its vapor can be in a state of dynamic equilibrium, i.e. the number of molecules emitted from a liquid is equal to the number of molecules returning from a vapor to a liquid, which means that the rates of evaporation and condensation are the same. This system is called two-phase . The vapor that is in equilibrium with its liquid is called saturated .
Saturated steam has maximum: pressure, concentration, density at a given temperature. They depend only on the temperature of the saturated steam, but not on its volume.
This means that if we had closed the vessel not with a lid, but with a piston, and after the steam had become saturated, we would compress it, then the pressure, density and concentration of steam would not change. More precisely, the pressure, density and concentration would increase for a short time, and the steam would become supersaturated. But immediately a part of the steam would turn into water, and the steam parameters would remain the same. If you raise the piston, the steam will no longer be saturated. However, due to evaporation, after some time it will become saturated again. It should be noted here that if there is no water at the bottom of the vessel or there is little of it, then this evaporation may be insufficient for the steam to become saturated again.
- The phrase: “In a closed vessel with water …” – means that saturated water is above the water.
- Dew drop means that the steam becomes saturated.
Absolute humidity ρ refers to the amount of water vapor contained in 1 m 3 of air (i.e. just the density of water vapor; the ratio of mass to volume is expressed from the Clapeyron-Mendeleev equation and the following formula is obtained):

where: p is the partial pressure of water vapor, M is the molar mass, R is the universal gas constant, T is the absolute temperature. The unit of measurement of absolute humidity in SI [ ρ ] = 1 kg / m 3 , although usually 1 g / m 3 is used .
Relative humidity φ is the ratio of absolute humidity ρ to the amount of water vapor ρ 0 that is necessary to saturate 1 m 3 of air at a given temperature:

Relative humidity can also be defined as the ratio of water vapor pressure p to saturated vapor pressure p 0 at a given temperature:

Evaporation can occur not only from the surface, but also in the volume of the liquid. In the liquid there are always tiny bubbles of gas. If the vapor pressure of the liquid is equal to or greater than the external pressure (that is, the gas pressure in the bubbles), the liquid will evaporate inside the bubbles. Bubbles filled with steam expand and float to the surface. This process is called boiling . Thus, the boiling of a liquid begins at a temperature at which the pressure of its saturated vapor becomes equal to the external pressure.
In particular, at normal atmospheric pressure, water boils at a temperature of 100 ° C. This means that at this temperature the pressure of saturated water vapor is 1 atm. It is important to know that the boiling point of the liquid depends on the pressure. In a hermetically sealed vessel, the liquid cannot boil, since at each temperature, an equilibrium is established between the liquid and its saturated vapor.
Surface tension
Molecules of a substance in a liquid state are located very close to each other. Unlike solid crystalline bodies, in which molecules form ordered structures in the entire volume of a crystal and can perform thermal vibrations around fixed centers, liquid molecules have greater freedom. Each fluid molecule, as well as in a solid, is “clamped” on all sides by neighboring molecules and performs thermal oscillations around a certain equilibrium position. However, from time to time, any molecule can jump abruptly to the next vacant place. Such jumps in liquids occur quite often; therefore, the molecules are not bound to certain centers, as in crystals, and can move throughout the entire volume of the liquid. This explains the fluidity of liquids.
Due to the dense packing of molecules, the compressibility of liquids, that is, the change in volume with pressure, is very small; it is tens and hundreds of thousands of times less than in gases.
The most interesting feature of liquids is the presence of a free surface. Liquid, unlike gases, does not fill the entire volume of the vessel in which it is poured. An interface forms between the liquid and the gas (or vapor), which is in special conditions compared to the rest of the liquid mass. Molecules in the boundary layer of a liquid, unlike molecules in its depth, are surrounded by other molecules of the same liquid not from all sides. The forces of intermolecular interaction acting on one of the molecules inside the fluid from neighboring molecules are, on average, mutually compensated. Any molecule in the boundary layer is attracted by molecules inside the fluid (the forces acting on this fluid molecule from the side of gas molecules (or vapor) can be neglected). As a result, some resultant force appears. directed deep into the liquid. If the molecule moves from the surface inside the liquid, the forces of intermolecular interaction will do a positive job. On the contrary, in order to pull a certain number of molecules from the depth of the liquid to the surface (that is, to increase the surface area of the liquid), it is necessary to expend the positive work of external forces ΔA external is proportional to the change in Δ S surface area.
Consequently, the molecules of the surface layer of the liquid have excess potential compared to the molecules inside the liquid. The potential energy E p of a liquid surface is proportional to its area:

The coefficient σ is called the surface tension coefficient ( σ > 0). Thus, the surface tension coefficient is equal to the work required to increase the surface area of the liquid by one at a constant temperature. In SI, the surface tension coefficient is measured in joules per square meter (J / m 2 ) or in newtons per meter (1 N / m = 1 J / m 2 ).
It is known from mechanics that the equilibrium states of a system correspond to the minimum value of its potential energy (any body always seeks to slide down a mountain, and not climb it). It follows that the free surface of the liquid tends to reduce its area. For this reason, the free drop of liquid takes a spherical shape. The fluid behaves as if forces acting on a tangent to its surface reduce (tighten) this surface. These forces are called surface tension forces . The presence of surface tension forces makes the surface of the liquid similar to an elastic stretched film. The surface tension force acting on a portion of the boundary of a fluid of length L is calculated by the formula:
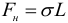
Thus, the surface tension coefficient σ can be defined as the modulus of the surface tension force acting per unit length of the line bounding the surface.
Capillary phenomena are called raising or lowering fluid in tubes of small diameter – capillaries. Wetting liquids rise through the capillaries, non-wetting drops. The height of the liquid column in the capillary :

When fully wetted θ = 0 °, cos θ = 1. In this case, the height of the liquid column in the capillary will be equal to:
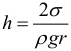
When fully nonwetting θ = 180 °, cos θ = –1 and, therefore, h <0. The level of non-wetting liquid in the capillary falls below the level of the liquid in the vessel into which the capillary is lowered.


