What is an ionic bond?
We explain what an ionic bond is and its different properties. In addition, its uses and some examples of this chemical bond.
-
What is an ionic bond?
It is understood by ionic bond or electrovalent bonding to one of the chemical binding mechanisms, which generally occurs between metallic and nonmetal atoms , fused due to the permanent transfer of electrons , and thus producing an electromagnetically charged molecule, known as an ion.
Electronic transfer in the ionic bond always occurs from metallic to non-metallic atoms, or in any case, from the most electronegative to the least. This is because the joint is produced by attraction between particles of different signs, whose variation in the coefficient of electronegativity is greater than or equal to 1.7 on the Pauling scale.
It should be clarified that although the ionic bond is usually distinguished from the covalent (consisting of a shared use of electronic pairs in the outer layer of both atoms), in reality there is no pure ionic bond , but this model consists of an exaggeration of the bond covalent , useful for the study of atomic behavior in these cases. But there is always some margin of covalence in these unions.
However, unlike the covalent bonds that often constitute polar molecules , ions do not have a positive and a negative pole, but only a single charge predominates in them . Thus, we will have cations when it is a positive charge (+) and we will have anions when it is a negative charge (-).
-
Properties of an ionic bond

The general characteristics of this type of link are:
- It is a strong link . Depending on the nature of the ions, the strength of this atomic junction can be very intense, so the structure of these compounds tends to form very resistant crystalline networks.
- It usually produces solids . At normal temperatures and pressure ranges , they usually produce crystalline and rigid molecular structure compounds, thus giving rise to salts. There are ionic liquids, too, or “melted salts,” which are rare but extremely useful.
- It has a high melting point . Both the melting point (between 300 ° C and 1000 ° C) and the boiling point of these compounds is usually very high, as large amounts of energy are required to break the electrical attraction between atoms.
- Water solubility . Most of the salts obtained in this way are soluble in water and other aqueous solutions that have an electric dipole (positive and negative poles).
- Electrical conduction . In their solid state they are not good conductors of electricity , since ions occupy very fixed positions in an electrical network. Instead, once dissolved in water or in aqueous solution, they become effective conductors of electricity.
- Selectivity . Ionic bonds can occur only between metals of groups I and II of the Periodic Table, and non-metals of groups VI and VII.
-
Ionic bond examples
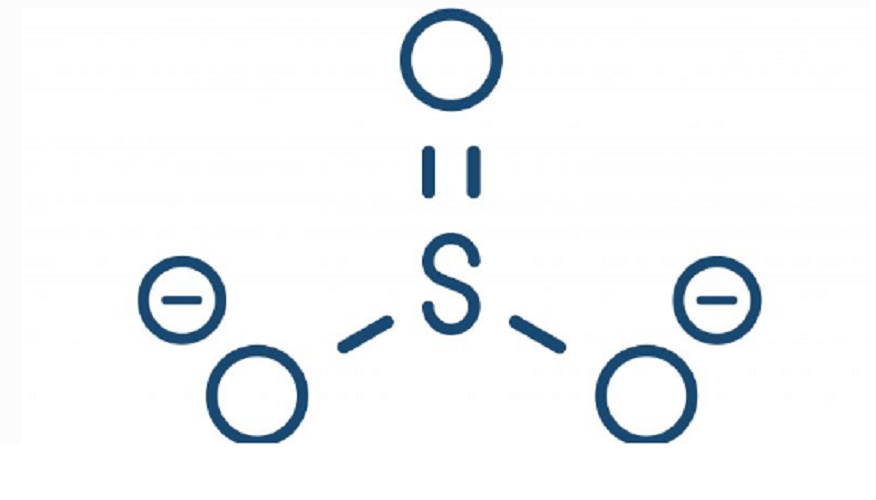
Some examples of ions obtained by this chemical process are:
- Fluorides (F – ) . Cathodic salts obtained from hydrofluoric acid (HF), used in the manufacture of toothpastes and other dental supplies.
- Sulfates (SO 4 2- ) . Salts or esters obtained from sulfuric acid (H2SO4), whose binding to a metal serves extremely diverse purposes, from additives in obtaining construction materials, to input for contrast radiographs.
- Nitrates (NO 3 -). Salts or esters obtained from nitric acid (HNO3), used in the manufacture of gunpowder (coupled with potassium), and in numerous chemical formulations for fertilizers or fertilizers.
- Mercury II (Hg +2 ) . It is a cation obtained from mercury, also called mercuric cation and which is only stable in acidic pH media (<2).
- Permanganates (MnO 4 -). The salts of permanganic acid (HMnO4), have an intense purple color and an enormous oxidizing power, which can be used in the synthesis of saccharin, for example, or in the treatment of wastewater , or in the manufacture of disinfectants.




