Types And Properties of Electric Charge With Simple Definition
We express the Types And Properties of Electric Charge, how it is classified and Also, what is Coulomb’s Law.
-
What is an electric charge?
In physics , a property of matter is called an electric charge . It is intrinsic to some subatomic particles and is evidenced by attractive or repulsive forces between them, through electromagnetic fields.
That is to say: matter can be electrically charged (gain charge), thus generating an electric field (or be charged precisely by an electric field with which it comes into contact), in what is known as electromagnetic interaction, one of the four fundamental interactions of nature .
An electric charge, then, is any matter that is charged with said electromagnetic force, and that is capable of transmitting it in some way, since they cannot be created or destroyed . However, not all matter responds in the same way to electromagnetic induction, and it depends on whether we talk about conductors ( electricity ) or insulators.
Under the International Measurement System (SI), electrical charges are measured in a unit called Coulombs or Coulombs (C) . Its name was established in honor of the French physicist Charles-Augustin de Coulomb (1736-1806), one of the greatest students of this type of physical phenomenon.
A Coulomb unit is defined as the amount of charge that an electric conductor passes through in one second, when the electric current is one ampere, and that corresponds to 6.242 x 10 18 free electrons.
Units
In the International System of Units the unit of electric charge is called coulomb or coulomb (symbol C). It is defined as the amount of charge that passes through the cross section of an electrical conductor in one second, when the electric current is one ampere , and corresponds to:
- 1 coulomb = 6,242 x 10 18 free electrons.
In Cegesimal System of Units (CGS) the electric charge of the electron,
- e = 4.8 × 10 -10 esu ( e lectro s tatic u nit , that is, electrostatic unit of charge) = 4.8 × 10 -10 statC
-
Types of electric charge

Electric charges interact with each other according to their type, which can be either positive or negative , as Benjamin Franklin called them. These denominations are arbitrary, that is, there is nothing that distinguishes the positive from the negative, but they are thought of just like a magnetic dipole, like a battery, which has a positive pole (or north) and a negative pole (or south).
The positive is denoted with the + sign and the negative with the – sign, conventionally. Electric charges of equal signs repel each other. Opposite sign electric charges attract each other .
-
Properties of electric charge
Electric charge is an intrinsic property of matter , and resides in subatomic particles : electrons are negatively charged and protons are positively charged (neutrons charge neutral, as the name suggests).
Thus, when an object is electrically charged, it is due to a shift in its electrons, either loss (gaining positive charge) or incorporation (gaining negative charge). Therefore, a material with an excess of electrons in its outer atoms will present a negative charge, while one with a deficit of them will present a positive charge.
In a closed system, the electric charge remains constant , just as it occurs within any chemical reaction.
Elemental electric charge
Current research in physics suggests that electric charge is a quantized property . The most elementary unit of charge was found to be the charge that the electron has , that is to say around 1,602 176 487 (40) × 10 -19 coulombs (C) and is known as elemental charge. 7 The value of the electric charge of a body, represented as q or Q , is measured according to the number of electrons it has in excess or in defect.
This property is known as quantization of the charge and the fundamental value corresponds to the value of electric charge that the electron has and which is represented as e . Any charge q that physically exists, can be written as where N is an integer, positive or negative.
By convention, the charge of the electron is represented as -e , for the proton + e and for the neutron, 0 . Particle physics postulates that the charge of quarks , particles that make up protons and neutrons take fractional values of this elementary charge. However, they have never observed free quark, and the value of its load together, in the case of the proton amount in the sum y + neutron 0.
Although we do not have a sufficiently complete explanation of why the load is a quantized quantity, which can only appear in multiples of the elementary load, various ideas have been proposed:
- Paul Dirac showed that if there is a magnetic monopole , the electric charge must be quantized.
- In the context of the Kaluza-Klein theory , Oskar Klein found that if the electromagnetic field was interpreted as a side effect of the curvature of a topology space-time , then the compactness of it would imply that the linear momentum according to the fifth dimension would be quantized and hence the quantization of the load was deduced.
In the International System of Units unit electric charge is called coulomb (symbol C) and is defined as the amount of charge to 1 meter exerts on another amount of charge equal force 9 x 10 9 N .
A coulomb corresponds to the charge of 6,241 509 × 10 18 electrons. 10 The value of the charge of the electron was determined between 1910 and 1917 by Robert Andrews Millikan and today its value in the International System according to the latest list of constants of the CODATA published is: 7
Since coulomb may not be manageable in some applications, as it is too large, its submultiples are also used:
- 1 millicoulomb =
- 1 microcoulomb =
The CGS system whose electric charge unit is the Franklin (Fr) is also frequently used . The value of the elemental load is then approximately 4,803 × 10 –10 Fr.
Load properties
Charge conservation principle
In agreement with the experimental results, the principle of conservation of the charge establishes that there is no destruction or net creation of electric charge, and states that in all electromagnetic processes the total charge of an isolated system is conserved.
In an electrification process , the total number of protons and electrons is not altered, there is only a separation of the electric charges. Therefore, there is no destruction or creation of electric charge, that is, the total charge is conserved. Electric charges may appear where there were none before, but they will always do so so that the total charge of the system remains constant. Furthermore, this conservation is local, it occurs in any region of space, however small it may be.
Like the other laws of conservation , the conservation of electric charge is associated with a Lagrangian symmetry , called quantum invariance in quantum physics . Thus by Noether’s theorem, each symmetry of the Lagrangian associated with a uniparameter group of transformations that leave the Lagrangian invariant corresponds to a conserved magnitude. 11 The conservation of charge implies, like the conservation of mass, that a continuity equation is satisfied at each point in space that relates the derivative of the density of electric charge to the divergence of the vector electric current density , said equation expresses that the net change in charge density{\ displaystyle \ rho} within a preset volume equals the integral of the density of electric current{\ displaystyle J} Over the surface that encloses the volume, which in turn is equal to the intensity of the electric current
Relativistic Invariant
Another property of electric charge is that it is a relativistic invariant . This means that all observers , regardless of their state of movement and speed , will always be able to measure the same amount of charge. 8 So, unlike the space , the time , the energy or momentum , when a body or particle moves at speeds comparable to the speed of light , the value of the load will not vary.
Electric charge density
Electric charge density is the amount of electric charge per unit length , area, or volume that is on a line , surface, or region of space, respectively. Therefore it is distinguished in these three types of charge density. 12 It would be represented by the Greek letters lambda (λ), for linear charge density, sigma (σ), for surface charge density and ro (ρ), for volumetric charge density.
There can be both positive and negative charge densities. Not to be confused with the density of charge carriers .
Despite the fact that the electrical charges are quantized with q and, therefore, multiples of an elementary charge, sometimes the electrical charges in a body are so close to each other that it can be assumed that they are uniformly distributed throughout the body of which they are part. The main characteristic of these bodies is that they can be studied as if they were continuous, which makes their treatment easier, without losing generality. Three types of electrical charge density are distinguished: linear, surface and volumetric .
Linear load density
It is used in linear bodies such as threads.
Where is the charge locked in the body and is the length. In the International System of Units (SI) it is measured in C / m ( coulombs per meter ).
Surface charge density
It is used for surfaces , eg a thin metal sheet such as paper aluminum.
where Q is the charge locked in the body and is the surface. In the SI it is measured in C / m 2 (coulombs per square meter ).
Volumetric charge density
It is used for bodies that have volume.
where is the charge locked in the body and the volume. In the SI it is measured in C / m 3 (coulombs per cubic meter ).
Ways To Change The Electric Charge of Bodies
Electrification is called the effect of gaining or losing electrical charges, usually electrons , produced by an electrically neutral body. The types of electrification are as follows:
- Electrification by contact: When we put a charged body in contact with a conductor, there can be a transfer of charge from one body to the other and thus the conductor is charged, positively if it “gave up electrons” or negatively if it “gained” them.
- Electrification by friction: When we rub an insulator with certain types of materials, some electrons are transferred from the insulator to the other material or vice versa, so that when both bodies separate they are left with opposite charges.
- Induction charge : If we approach a negatively charged body to an isolated conductor, the repulsive force between the charged body and the valence electrons on the surface of the conductor causes them to move to the part furthest from the conductor to the charged body, leaving the closest region with a positive charge, which is noted by having an attraction between the charged body and this part of the conductor. However, the net conductor load remains zero (neutral).
- Charging by the photoelectric effect : It happens when electrons are released on the surface of a conductor when irradiated by light or other electromagnetic radiation.
- Charge by electrolysis : Chemical decomposition of a substance, produced by the passage of a continuous electric current.
- Charging by thermoelectric effect : It means producing electricity by the action of heat.
-
Coulomb law
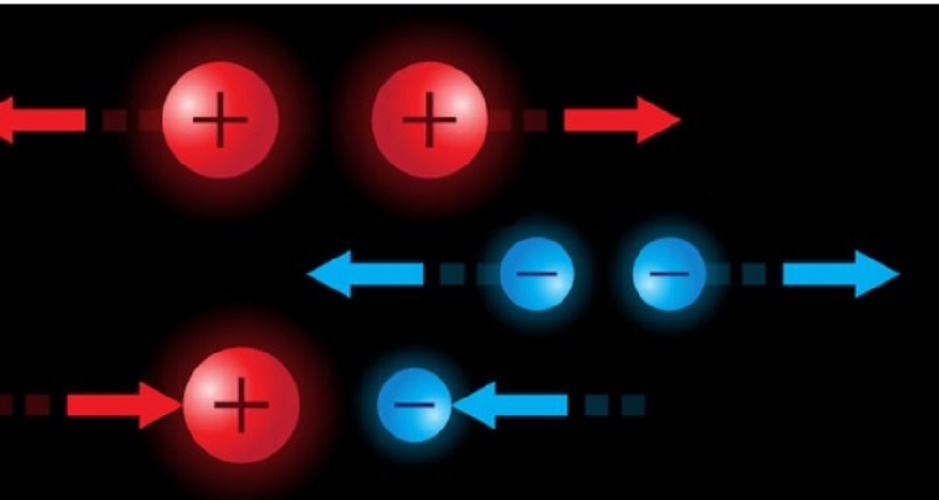
Coulomb’s Law or Fundamental Principle of electrostatics governs the magnitude of the attractive or repulsive forces between two determined electric charges , regardless of the sign of their charge (obviously, if they are of the same sign, it will be a repulsive force; and if they are of opposite sign, of attraction).
According to this law, said force is proportional to the product of the value of the charges, and inversely proportional to the square of the distance that separates them. This is expressed mathematically in the following way:
F = K. [(Q1 x Q2) / r 2 ]
Where:
- F is the force.
- Q are the two respective charges.
- r is the distance that separates them.
- K is a proportionality constant defined as 9.10 9 Nm 2 / C 2 .
















