Bohr Atomic model Postulates – WhatMaster
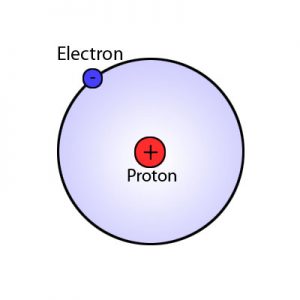
The atomic model of Niels Bohr shows the atom as a small positive nucleus surrounded by electrons in circular layers around the nucleus.
With the help of Plank’s quantum theory, the light spectra of the elements, and Rutherford’s nuclear theory, Bohr succeeded in 1913 in establishing a new atomic model where electrons described circles around the nucleus.
This model served to explain the spectra of light emitted by the elements and the regularities of the periodic table. In addition, with the Bohr model, the quantum age began.
Postulates of the Bohr atomic model
The electrons move at a certain distance from the nucleus
For Bohr, the circular motion of the electron had a specific radius of rotation, so there could be no electrons between two layers.
The electrons move stably, that is, without releasing energy, in the steady-state. When an amount of external energy is applied to them, that is, the electrons are excited, they can jump to a higher energy level. This is the least stable excited state of the electron.
The electrons follow a circular path
The electrons revolve around the nucleus in defined orbits.
The electrons in steady-state move in “energy levels” or “layers”. Each level of energy is called by letters, the lowest level being the letter K, followed by L, M, N, and so on. The layers can be imagined as onion rings enveloping the nucleus.
In this sense, the layers closest to the nucleus have less energy, Also each layer could contain more than one electron, for example, the K layer can have up to two electrons, the L layer, eight, and so on.
Electrons emit light when they change levels
All elements when heated emit a light of specific colors or frequencies, known as the electromagnetic spectrum.
When an electron jumps from a high energy level to a lower energy level, the energy difference is released in the form of electromagnetic radiation or light. Thus, the energy is related to the frequency or color of the light fby the Max Planck relation h : Energy = hf .
Characteristics of the Bohr model
- The electrons describe circular orbits around the nucleus.
- Electrons travel at predetermined levels of energy.
- Electrons can jump from a lower energy level to a higher energy level if it provides them with energy.
- When the electrons return to their stationary energy level, they release light.
How did Bohr reach the atomic model?
Niels Bohr was born in Copenhagen (Denmark) in 1885. In 1912 he entered the laboratory of Ernest Rutherford with the task of discovering the way in which the nuclear structure of the atom was stable.
The atomic model of Rutherford was based on a central nucleus surrounded by electrons, as in the solar system. This simple model had a problem: an electron spinning around the nucleus had to emit electromagnetic radiation, with the consequent loss of energy. This would cause the electron to fall into the nucleus, causing the atomic collapse.
Bohr proposed that the electrons were at a certain distance from the nucleus, spinning in circles. Each circle or orbit was provided with a predetermined amount of energy. If an electron jumped into an orbit of lower energy, it would emit electromagnetic radiation.
Failures of the Bohr atomic model
At first, the Bohr model seemed promising. It fitted perfectly to the hydrogen atom. But when it came to applying this model to other atoms, it did not work.
Electrons do not move in circular orbits
Some tried to fit the model to an elliptical orbit, without success. Nowadays it is known that the electron behaves like a wave and a particle, so it is better to speak of a wave function, a space around the nucleus where there is a probability of finding the electron.
Related Links




